What exactly is hard & soft water?
The terms hard water and soft water are used to describe water containing high (hard) or low (soft) levels of dissolved minerals.
Primarily, these minerals are magnesium and calcium.
Hard water:
- Hard water contains high levels of magnesium, calcium, iron, manganese, or zinc, and has a mineral-rich taste that many people prefer.
- Hard water also leaves mineral scale in pipes and on kitchenware, which can reduce appliance performance and cloud drinking glasses.
- Hard water is more common in groundwater sources. That’s why many domestic well owners need to use a water softener, or a softener & filter combination in a well water filtration system, to reduce their water hardness.
Soft water:
- Soft water contains low levels of the same minerals and has a milder taste as a result.
- Soft water feels softer on skin and hair and allows soaps to lather more easily for efficient showering and laundry.
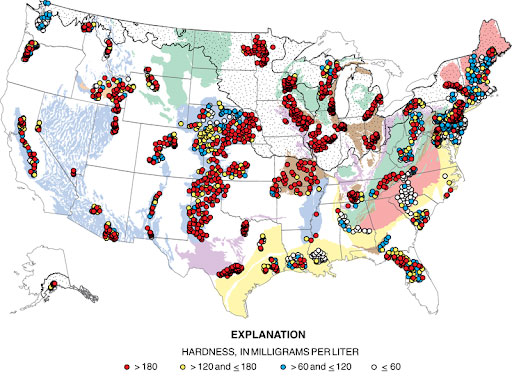
map of hard water in the USA
In this article, we’ll go through full explanations of hard water and soft water, plus the things you can do to soften water in your home.
Scroll to:
- What is hard water?
- What is soft water?
- Do I have hard water or soft water?
- What does a water softener do?
What is hard water?
Hard water is water from a natural source with high calcium, magnesium, or other mineral content. Scientific definitions of water hardness are often based on the amount of dissolved calcium carbonate. For example, the U.S. Geological Survey classifies hard water as follows:
- Moderately hard water – 61 to 120 mg of calcium carbonate per liter of water,
- Hard water – 121 to 180 mg of calcium carbonate per liter,
- Very hard water – over 180 mg of calcium carbonate per liter.
However, the general public often judges water hardness by taste, feel, and appearance, rather than specific mineral content. Either way, not all mineral water is considered hard.
What is hard water good for?
Drinking hard water won’t fulfill recommended daily requirements of essential minerals like iron, calcium, and magnesium, but it can definitely contribute. These minerals can also contribute to better-tasting water.
What are the downsides of hard water?
Hard water can leave residues on dishes, glasses, and faucets. Often called “scale”, this can be a particular problem in appliances that heat water, which increases the rate of calcium deposit formation.
Washing machines, dishwashers, kettle, coffee makers, and water heaters are all vulnerable to collecting scale. As the minerals build up, appliance performance can be significantly affected. This includes inside pipes, where scale can reduce efficiency and increase energy bills.
Hard water is often less pleasant to shower and wash with. It can make hair feel scratchy and makes it more difficult to lather soaps.
Where is hard water found?
As groundwater moves through soil and rock, minerals and metals become dissolved into it. That’s why well water is often much harder than water from lakes and reservoirs. If the soil around bodies of surface water is particularly calcium-rich, however then that water is also likely to become hard.
When the USGS surveyed groundwater from over 2000 domestic wells across the US, they found calcium-rich soil and hard water is prevalent across the country, but particularly on the East Coast and in the Midwest.

map of hard water in the USA
What is soft water?
Soft water contains low levels of calcium and magnesium. This means that it doesn’t produce any of the hard water effects caused by those minerals.
What is soft water good for?
Most people prefer soft water for bathing, thanks to its softer feel on hair and skin. Soft water is also preferable for cleaning since it creates more suds and is less likely to produce mineral stains or deposits.
Because it doesn’t leave scale, using soft water in your home can increase the performance and lifespan of hot water devices like dishwashers and heaters.
What are the downsides of soft water?
Soft water can have a filmy or slippery feel that some people don’t like. It may also have a milder taste—or even be tasteless—due to a lack of mineral content. Soft water doesn’t impart any of the minor health benefits that drinking mineral water does.
Where is soft water found?
Rainwater is inherently soft and only becomes harder once it spends under the earth. Water will take on minerals and contaminants at different rates depending on their prevalence in the local region.
As the map above indicates, soft water is found more on the West than East Coast, though it’s also common in New England and other areas with a lot of granite bedrock, which isn’t very porous so won’t impart as many minerals.
Do I have hard water or soft water?
Most areas of the US have hard water, so it’s a safe bet that your water supply registers somewhere between moderately hard and very hard according to the USGS criteria.
If you’re not sure whether you have hard or soft water, here’s a simple DIY home test: fill a bottle with tap water and a few drops of liquid soap. Shake the bottle and observe how many suds are created. The fewer the suds, the harder the water.
Extremely hard water produces almost no suds, instead of turning into a cloudy solution:
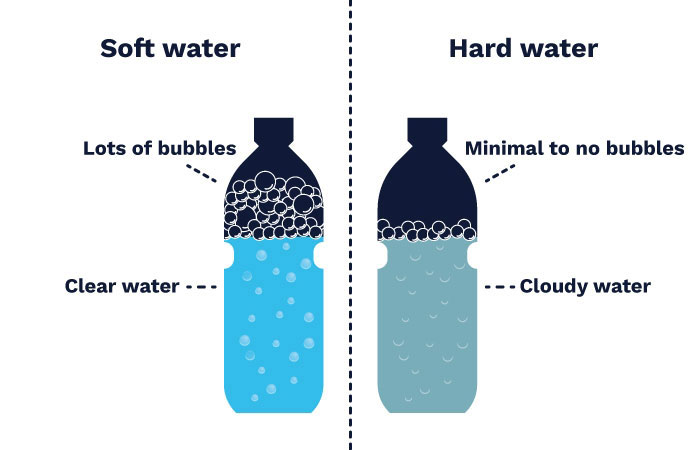
hard water vs soft water comparison
Check with your water provider
If you’re on city water, a simple method for estimating your water hardness is to just ask your water supplier or local government. Often, they keep up-to-date data on water hardness and can provide you with the info you need.
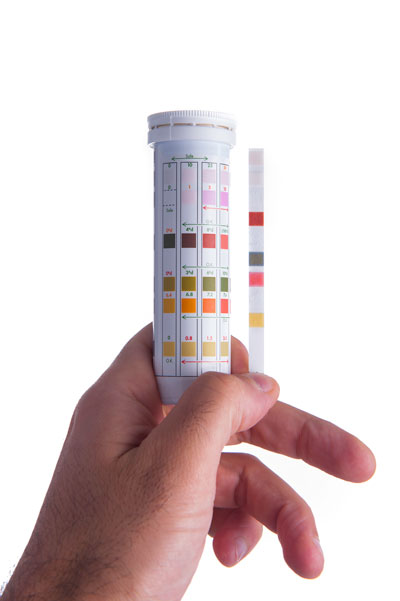
Hard water test kits
To go a step above the suds bottle test, purchase a hard water kit from a home improvement store or register for a test through an independent lab. Water testing can also reveal the presence of other unwanted chemical or bacterial contaminants, especially in well water.
hard water test strip set. Test strips have small amounts of chemicals on them that react to the minerals in hard water. Some manufacturers make different strips for testing drinking water and aquariums, so make sure you buy the right type for your purpose.
To use a test strip, simply dip the designated end into a glass or bowl of water and hold it there for about a second, until the end changes color. You shouldn’t have to hold it in the water for long, so pull it out once you have your reading.
You can also take a few test strips to different parts of your home, to see if water hardness varies in one part of your house or another.
Once you have completed the test, compare the color on the tested strip to the legend or explanatory scale that came with the kit to get a precise estimate of your water hardness. Simply match the color on the end of the strip to the closest color on the given scale. Usually, a darker color means harder water.
7 signs of hard water
- Dry skin or hair – your skin is itchy and your hair is dull and flat.
- Foggy or dirty dishes – Dishes rinsed in hard water will have spotty, chalky spots.
- Laundry issues – Your clothes will be stiff or scratchy because hard water makes it hard to rinse laundry detergent.
- Soap scum – Soap will cling and form crusts on your showerhead, bathtub, and sink.
- Appliance damage – Your water-based appliances will likely have limescale or calcium mineral deposits and you may notice your laundry machine or dishwasher breaking.
- Low water pressure – Water flowing from your tap will likely clog over time and not run as smoothly or steadily.
- Sour or metallic taste – As you drink from your tap you may notice that it tastes sour and sulfuric or smells metallic.
What does a water softener do?
To reduce water hardness and the unwanted effects of hard minerals, water softeners use one of two technologies: ion exchange and water conditioning.
Ion exchange softeners
Calcium and magnesium are composed of positively charged ions, which means they easily bond with other metals and minerals (such as the inside of pipes and water heaters).
Ion exchange water softeners take advantage of this property to attract hard minerals and exchange them for inert compounds that don’t cause scale.
Most softeners use a resin coated with sodium (and/or potassium) ions, which have a negative charge. As water passes over the resin, the negatively charged sodium ions are attracted to and exchanged with the positively charged hard mineral ions in the water.
Once the negatively charged resin bed becomes saturated, the softener enters a regeneration phase, which lasts 2-3 hours and is usually programmed to occur overnight.
Salt-free water conditioners
Instead of removing and replacing hard minerals, water conditioners reformat the composition of hard water so that minerals can’t bind with exterior surfaces and cause scale.
Most conditioners use a crystallization process to change the chemical makeup of water. As calcium and magnesium ions pass through the softener, they stick to tiny “nucleation” sites in the device media, where they form microcrystals.
In this crystallized form, the mineral ions are less likely to attach to pipes and appliances and may even attract existing scale. Because they don’t remove hard minerals, salt-free conditioners don’t require a regeneration phase.
We reviewed the best salt-free water softeners on the market right now. Check out the results here.
3 water softener benefits
- Money-saving – The more mineral build-up exists in hot water appliances, the harder they have to work, and the more energy they use. Using a water softener may mean savings on plumbing, electric bills, and even soaps and detergents.
- Helps with cleaning – Hard water is more likely to clog and create stains or scum. Soft water means less time re-washing dishes or putting in laborious hours to scrub your sink and tub.
- Soft hair and skin – Hard water is more likely to strip skin and hair of natural oils. Softer water can help hold moisture in the skin and retain pH hair’s balance.
Can standard water filters soften water?
Brita pitchers and other common devices filter water using carbon cartridges, which attract and bind organic contaminants such as calcium and magnesium in water. This means that they can reduce hard minerals and make a noticeable difference in water taste.
However, these devices don’t have the capacity or density to soften water to the same degree as water softeners—meaning that calcium and magnesium are likely to remain after filtering. To achieve filtration and softening with a single device, a water filter and softener combo system is required. Read our list of the best whole house water filter and softener combos.
Summary
- Calcium and Magnesium deposits turn soft rainwater into hard groundwater.
- Hard water is more likely to occur in regions with porous bedrock, such as sandstone or limestone.
- There may be minor health benefits to drinking mineral-rich hard water.
- Hard water is bad for hot water appliances, as it leaves mineral deposits that build over time and interfere with functioning.
- Soft water is has a more pleasant feel for showering.
- Water softeners can effectively reduce the amount of calcium and magnesium in water through a process called ion exchange.

